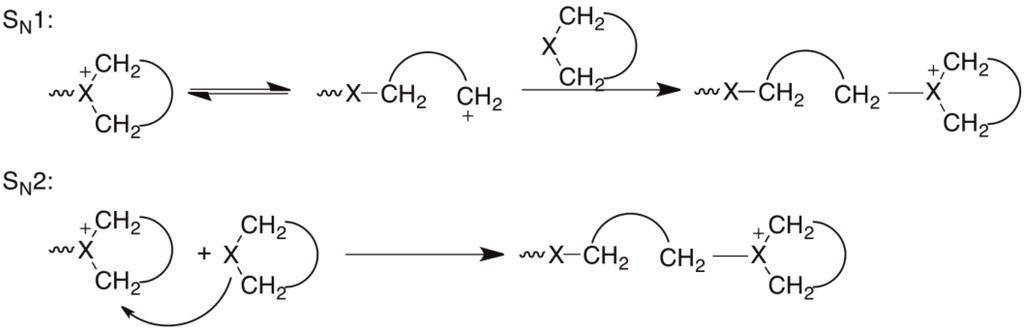
Radical Ring-Opening Polymerization: Scope, Limitations, and Application to (Bio)Degradable Materials | Chemical Reviews

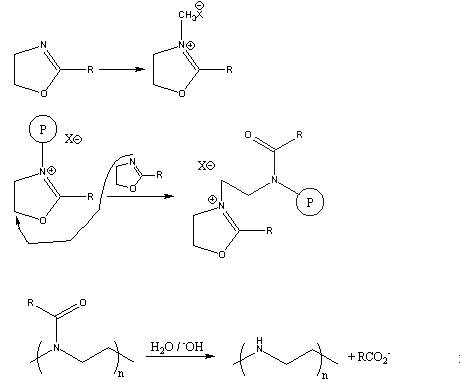
Mechanism of cationic ring-opening polymerization of 2oxazolines. R can... | Download Scientific Diagram

Ring‐opening polymerization of ϵ‐caprolactam and ϵ‐caprolactone via microwave irradiation - Fang - 2002 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

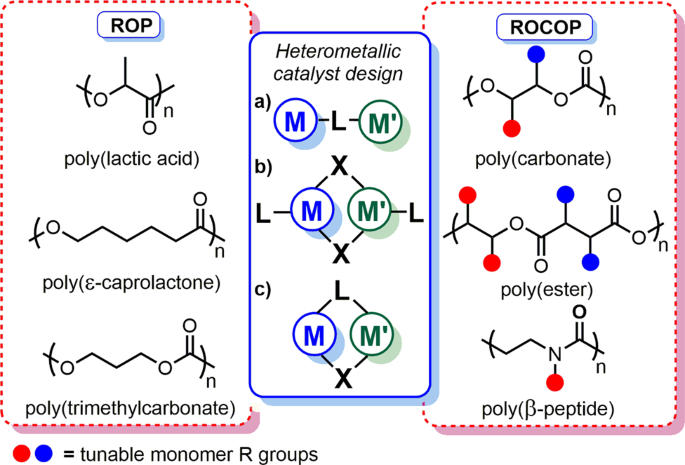
Ring opening polymerization of unsubstituted lactones, lactides, and... | Download Scientific Diagram
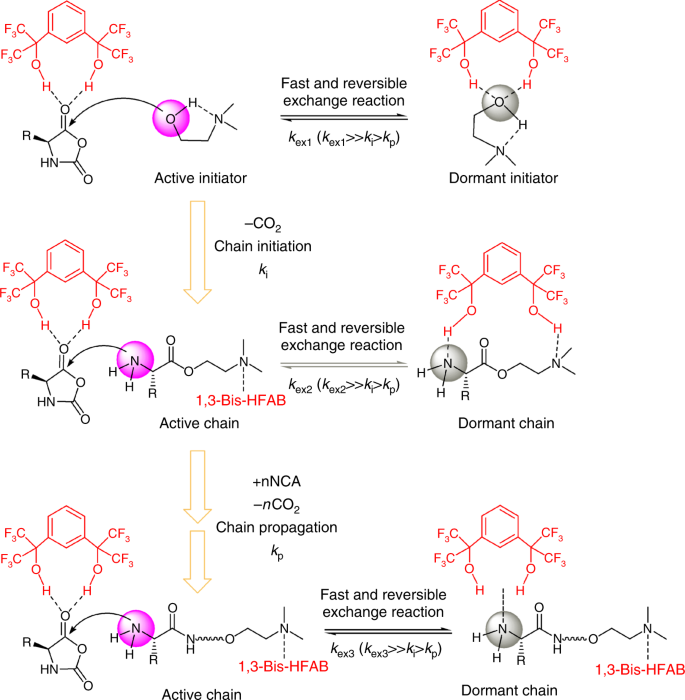
Fast and selective organocatalytic ring-opening polymerization by fluorinated alcohol without a cocatalyst | Nature Communications

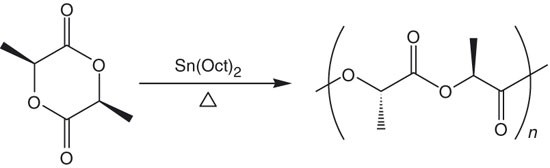
Coordination Insertion Mechanism of Ring‐Opening Polymerization of Lactide Catalyzed by Stannous Octoate† - Rao - 2021 - Chinese Journal of Chemistry - Wiley Online Library
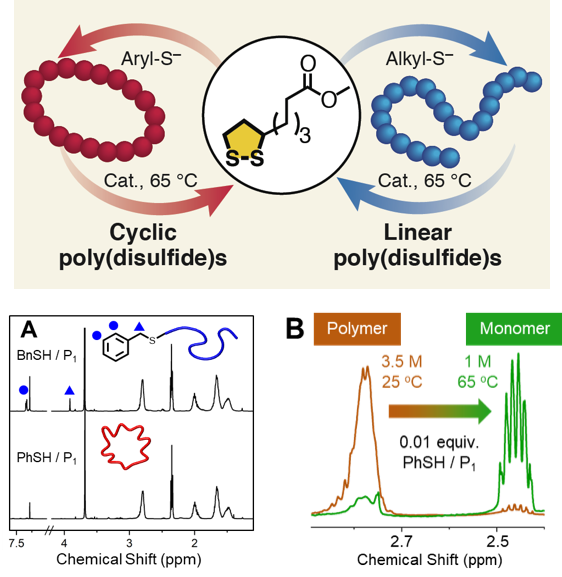
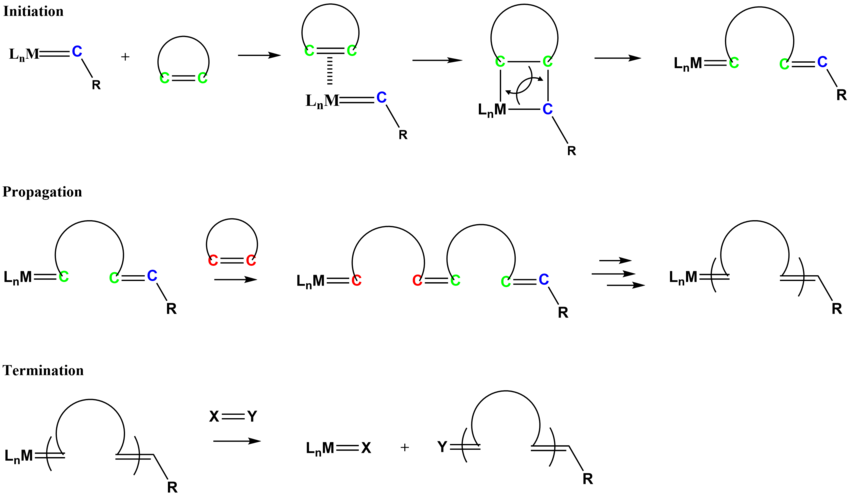
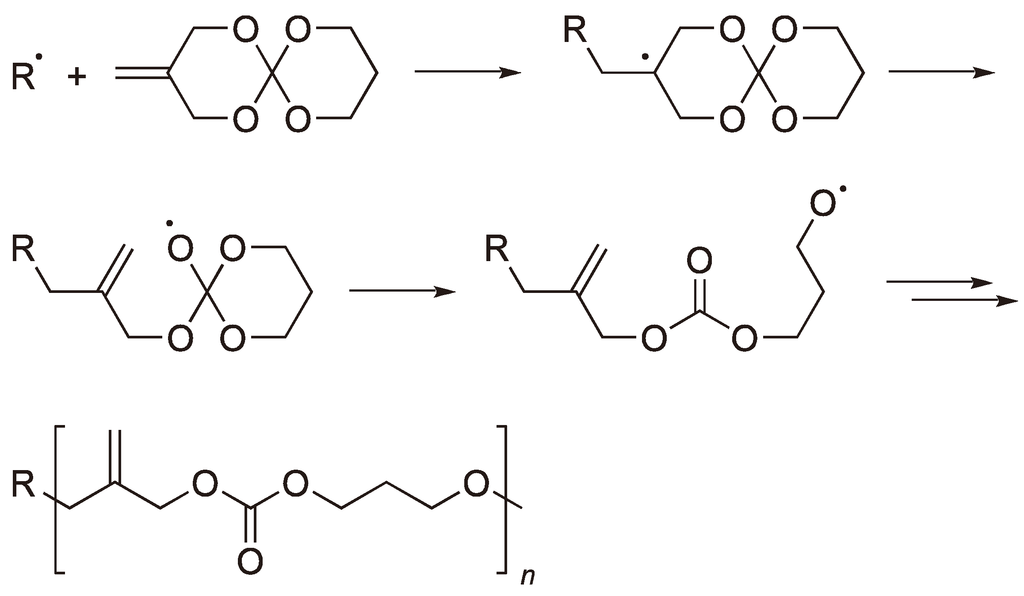
Architecture-Controlled Ring-Opening Polymerization for Dynamic Covalent Poly(disulfide)s - Joint Center for Energy Storage Research

Ring-opening polymerization of cyclic 1,3-oxathiolane-2-thione promoted by neighboring sulfide group and ring contraction - ScienceDirect
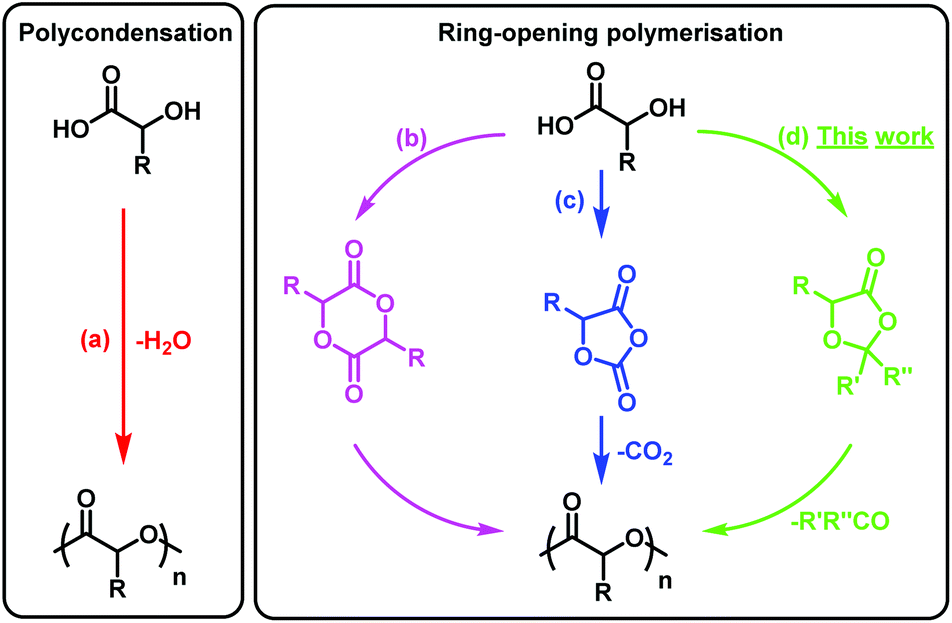
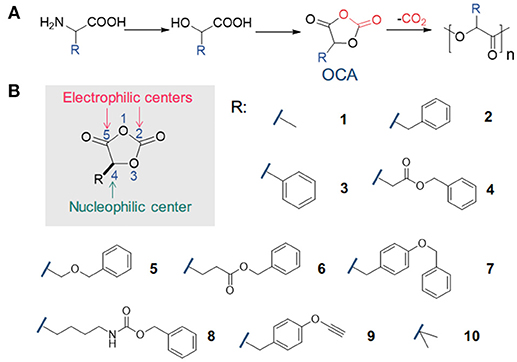
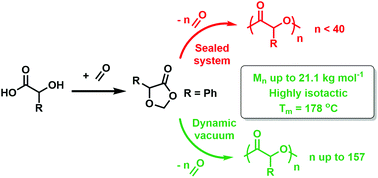
Understanding the ring-opening polymerisation of dioxolanones - Polymer Chemistry (RSC Publishing) DOI:10.1039/C8PY01695J

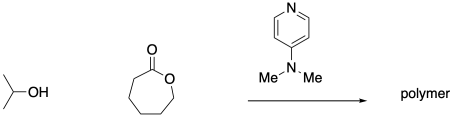
Ring-opening polymerization of ɛ-caprolactone by poly(propyleneglycol) in the presence of a monomer activator - ScienceDirect