Biosynthesis of ebelactone A: isotopic tracer, advanced precursor and genetic studies reveal a thioesterase-independent cyclization to give a polyketide β-lactone | The Journal of Antibiotics

Size range of lactone rings produced by TE domains. The lactone ring of... | Download Scientific Diagram
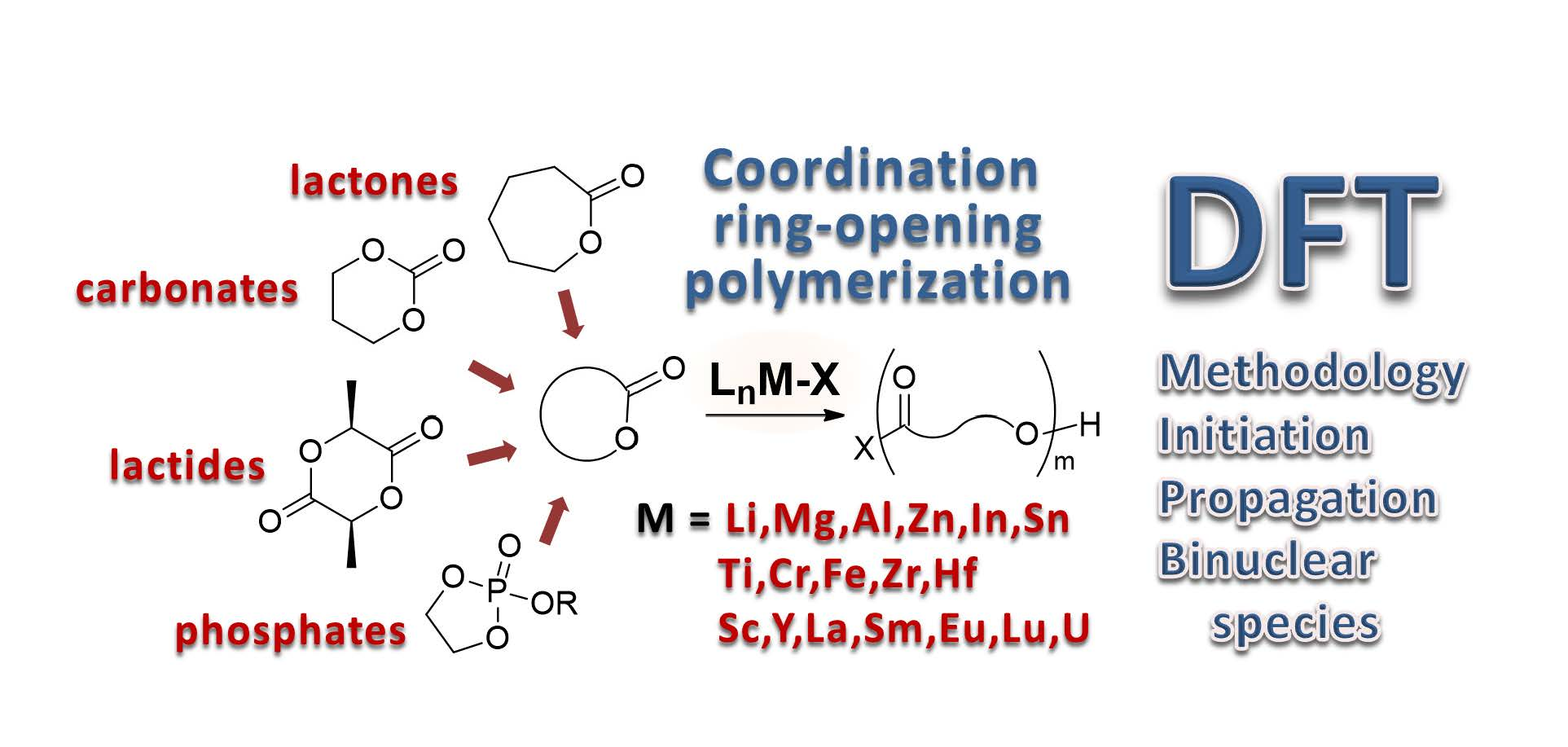
Molecules | Free Full-Text | Coordination Ring-Opening Polymerization of Cyclic Esters: A Critical Overview of DFT Modeling and Visualization of the Reaction Mechanisms

Synthesis of New Camptothecin Analogues with the E‐Lactone Ring Replaced by α,β‐Cyclohexenone - Bacherikov - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry

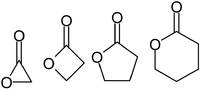
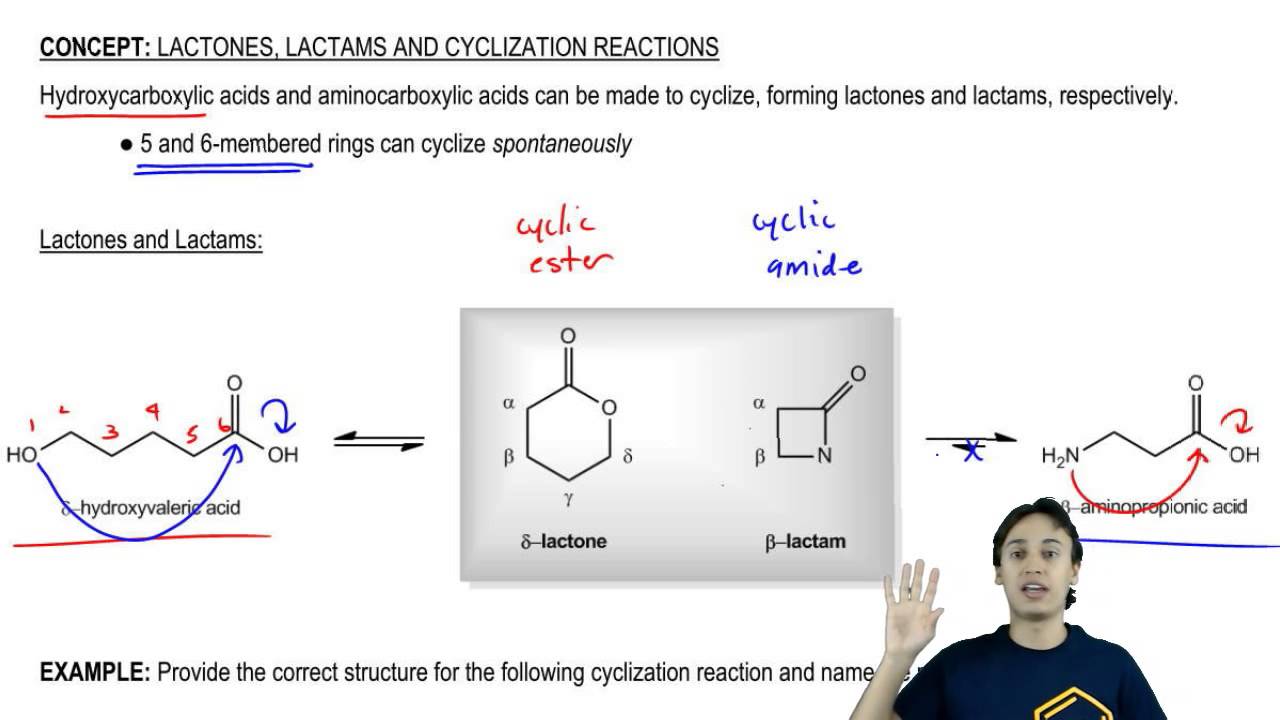
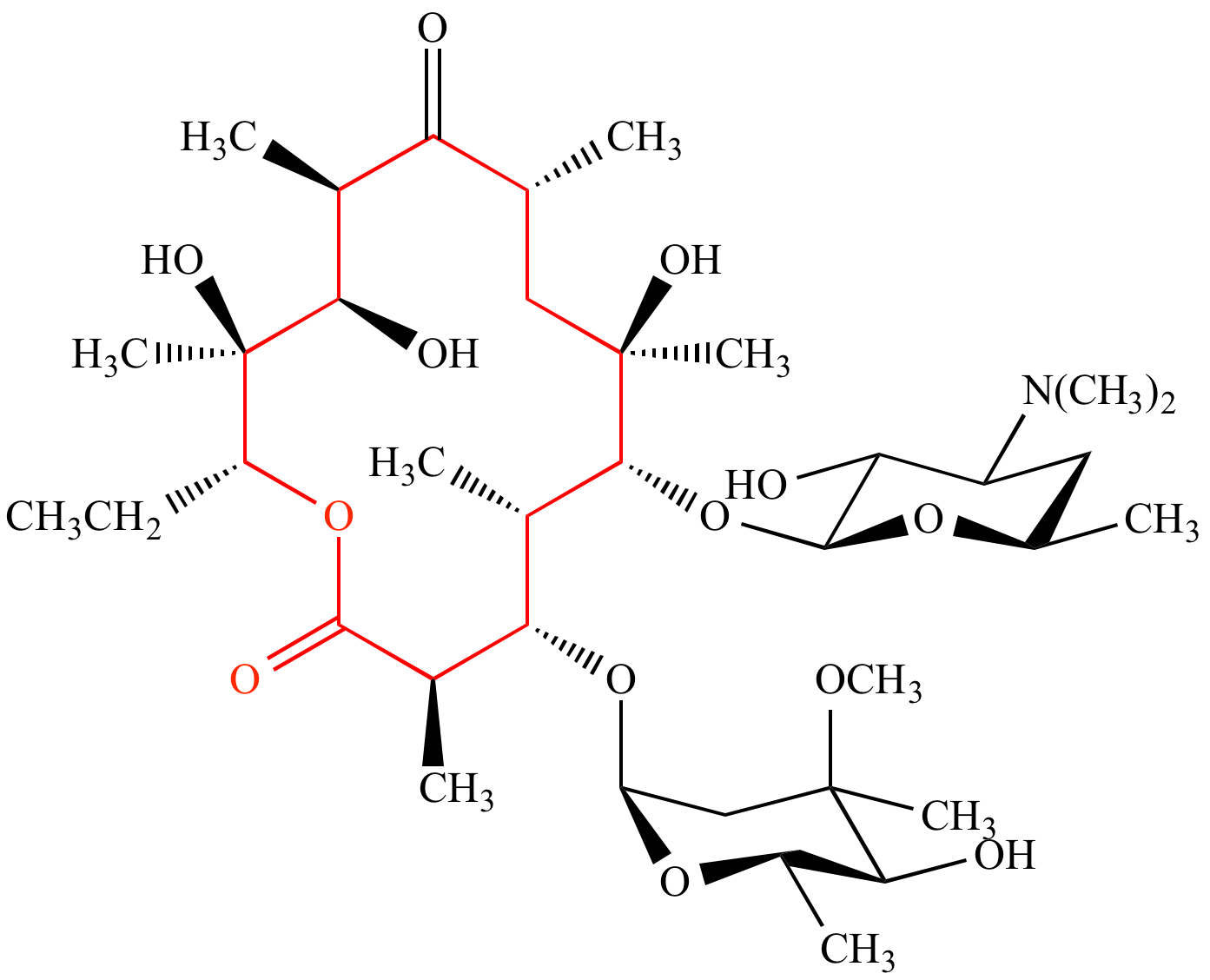
Lactones: Classification, synthesis, biological activities, and industrial applications - ScienceDirect

the Sugar and Lactone Ring Structure-Activity Relationships for the Hypertensinogenic Activity of Ouabain : Role of | Semantic Scholar
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE

Controlled Random Terpolymerization of β-Propiolactone, Epoxides, and CO2 via Regioselective Lactone Ring Opening | CCS Chem